|
This is a mirror page of the Inoceramid pages of H. Hilbrecht |
|
Lower Turonian Euramerican Inoceramidae: A morphologic,
taxonomic, and biostratigraphic overview
A report from the first Workshop on Early Turonian
Inoceramids (Oct. 5-8, 1992) in Hamburg, Germany;
organized by Heinz Hilbrecht and Peter J. Harries
by Peter J. HARRIES (Tampa, FL, USA), Erle G. KAUFFMAN
(Boulder, CO, USA), James S. CRAMPTON (Lower Hutt, NZ)
(Redacteurs),
and P. BENGTSON (Heidelberg, Germany), S. CECH, (Prague,
Czech Republic), J. A. CRAME (Cambridge, UK), A. V. DHONDT
(Brussels, Belgium), G. ERNST (Berlin, Germany), H.
HILBRECHT (Zürich, Switzerland), G. LOPEZ (Barcelona,
Spain), R. MORTIMORE (Brighton, UK), K.-A. TRöGER
(Freiberg, Germany), I. WALASZCZYK (Warsaw, Poland), and C.
J. WOOD (Croydon, UK).
-
Reference:
Harries, P.J., Kauffman, E.G., Crampton, J.S. (Redacteurs), Bengtson, P., Cech, S., Crame, J.A., Dhondt, A.V., Ernst, G., Hilbrecht, H., Lopez, Mortimore, G.R., Tröger, K.-A., Walaszcyk, I. & Wood, C.J. (1996): Mitteilungen aus dem Geologisch - Paläontologischen Museum der Universität Hamburg, 77: 641-671.
INTRODUCTION
The bivalve family Inoceramidae first evolved in the Permian and became extinct at the close of the Mesozoic. Inoceramids became dominant elements of many level-bottom communities, and they achieved global dispersion during the Jurassic and Cretaceous, especially during intervals of restricted benthic oxygen and black-shale deposition. Many groups appear to have been specifically adapted, in terms of anatomy and morphology (Kauffman and Harries, 1992), and possibly through chemosymbioses (Kauffman, 1988b; MacLeod and Hoppe, 1992), to chemically deleterious benthic environments. They are also found, however, in a wide range of different facies types/environments (from basinal black shales to nearshore sands), which suggests that they had relatively wide ecological tolerances at the genus and species level. The majority of inoceramid species had intercontinental or cosmopolitan distribution, mirroring the widespread nature of their preferred habitats, their broad adaptive ranges, and probably long-lived planktotrophic larvae; large larval shells are known from a few species. Despite their broad distribution, the Inoceramidae appear to have evolved very rapidly, with species ranges commonly averaging 0.2-0.5 Ma. This greatly enhances their use in biostratigraphy, and contrasts to the "normal" evolutionary rates of bivalves (2 Ma; see Stanley, 1979) and to the evolutionary hypothesis that cosmopolitan taxa should have slow evolutionary rates because of the wide dispersion of their component populations.
Despite the fact that the Inoceramidae have been studied intensively for over a century, there are still a wide range of problems remaining to be investigated. The Lower Turonian, although only a brief portion of inoceramid history, represents an ideal interval to study a number of these problems which can, at least in concept, be applied to many different time periods as well. These include: 1) the effects of a mass extinction (Cenomanian-Turonian) on inoceramids; 2) a switch in generic dominance from Late Cenomanian Inoceramus s.s. to Early Turonian Mytiloides within the same basic facies; 3) the unusually rapid evolutionary rates of Cenomanian-Turonian inoceramids at a time when species were achieving their widest dispersal (a seeming contradiction); 4) the need for generic revision of the Inoceramidae in this interval; 5) problems with species-level taxonomy related to the seeming morphologic plasticity of the forms; and 6) the extremely similar forms of Early Turonian Mytiloides spp. to those which evolved in the Late Turonian to basal to middle Coniacian, possibly representing iterative evolution. This overview paper will focus on the morphological terminology required to describe most inoceramids, the techniques of biometric analyses, concentrating on shape analysis, that can be employed in inoceramid studies to resolve problems of population variation in evolutionary and taxonomic studies of the Inoceramidae, generic diagnoses for all known Lower Turonian inoceramid genera, and attempts to construct a Lower Turonian inoceramid biostratigraphy.
This report focuses on many, but not all, of the topics discussed at the Hamburg workshop. Whereas most of the taxonomic, morphologic, morphometric, ecologic, and broad biostratigraphic aspects of this report represents a consensus of the participants and co-authors, differences of opinion naturally arose during our discussions and subsequent manuscript reviews. Points of major debate and differing opinions are noted throughout the manuscript, which we intend as a "state-of-the-science, 1993" statement designed to stimulate further research and discussion. Harries, Kauffman, and Crampton are solely responsible for designing the format and compiling the initial draft of the manuscript, as agreed upon by the meeting participants. The present paper, however, incorporates many observations, changes, and deletions suggested by the workshop participants during the meeting and manuscript review process. The Redacteurs are grateful for the constructive suggestions of our colleagues.
MORPHOLOGIC FEATURES OF INOCERAMID SHELLS
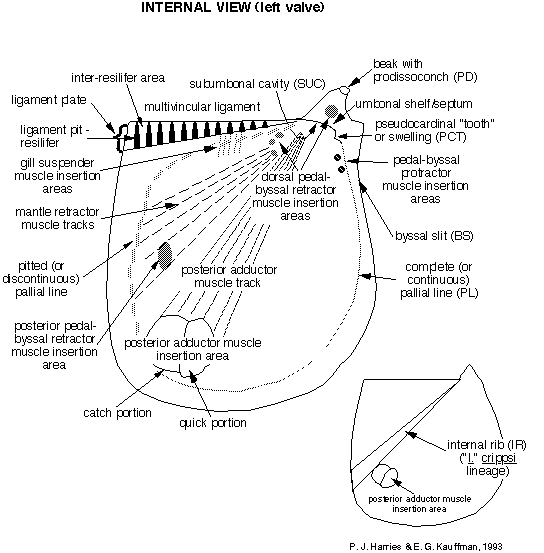
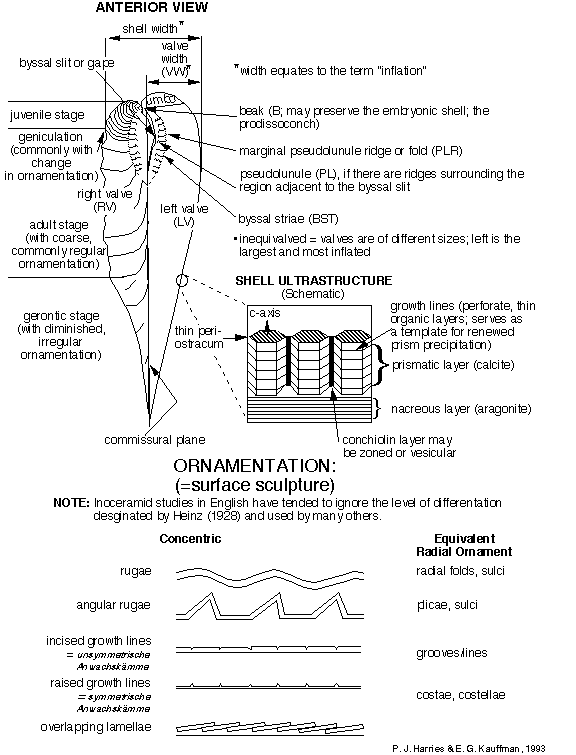
Throughout the history of inoceramid research, there have been a wide variety of different morphologic features and various morphometric parameters employed to make species determinations. A comprehensive, nomenclaturally consistent system for defining morphologic features of the inoceramid shell is essential to morphometric, functional morphologic, and evolutionary studies, as well as anatomical reconstructions, within the family. External shell morphology, upon which the great majority of taxa are based, has been well-defined. However, it is complicated by iterative or parallel evolution among only distantly related groups of inoceramids and even their ancestors. A knowledge of internal shell features have become critical for taxonomic (especially generic) determination, although for many species musculature and ligamenture are only poorly known or not yet documented. Utilization of both internal and external shell features in the Inoceramidae will help to maintain a degree of constancy in descriptive format and systematic concepts. Figure 1 , Figure 2 , and Figure 3 , compiled by P. J. Harries and E. G. Kauffman, illustrate the key morphologic features used today in systematic description of most inoceramids. A glossary of these terms is presented in Appendix A. The simplest, most descriptive and most commonly used English language terminology is utilized in this glossary, with equivalent German language terms, where relevant, listed in italics within parentheses. But the reader should be aware that this morphologic review draws heavily from earlier work by Heinz (1932), Seitz (1934), Dobrov and Pavlova (1959), Tröger (1967), Kauffman and Powell (1977), Efremova (1978), Keller (1982), and Rasemann (1986) and is a compilation of terminology for internal and external shell features, as well as for shell ultrastructure, used by a variety of previous authors. Some new terminology has been added by the authors.
BIOMETRIC ANALYSIS OF INOCERAMID BIVALVES
Inoceramid taxonomy is hampered by two general problems. First, for genetic and taphonomic reasons, the inoceramid taxonomist typically has relatively few characters to utilize in taxonomic differentiation, and existing classifications are based almost exclusively on shape and external shell features. Secondly, inoceramids commonly display considerable intraspecific (phenotypic, ecophenotypic, ontogenetic) morphological variation, and interspecific morphological convergence in their evolution. For these reasons, an increasing number of workers have attempted to use quantitative biometric methods as objective aids in the classification of the group. Two general classes of methods are discussed below: 1) those based on the uni- and bivariate analysis of distance and angle measurements and 2) multivariate outline-shape analysis.
Univariate and bivariate methods
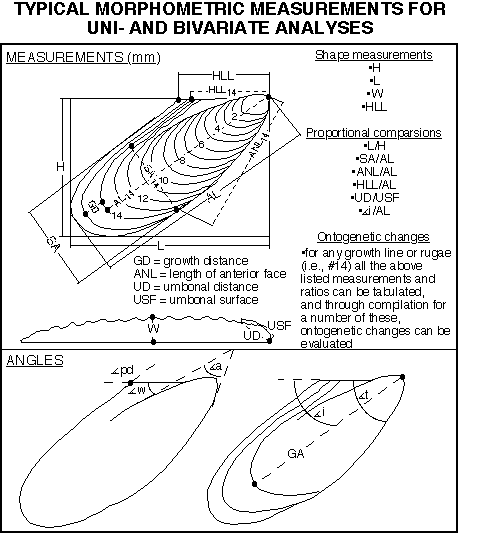
Distance and angle measurements have been used extensively to describe inoceramid shape and ornament in order to quantify intra- and interspecific variation. A great number of measurements have been used, a reflection of the diversity of inoceramid morphologies which probably precludes definition of a universally applicable set of parameters. Many of the morphological elements measured have been reviewed by Sornay (1966), Efremova (1978), Rasemann (1986), and Aliev et al. (1988). Figure 4 shows a number of measurements which have been used to describe Turonian Mytiloides and similar forms.
Distance and angle measurements have typically been examined and compared using standard univariate statistics and bivariate plots (e.g., see Jones 1988 for a review of some useful uni- and bivariate statistics). By taking measurements from different growth stages of single individuals some studies have examined patterns of relative growth, or changes of shape occurring throughout the ontogeny of an individual. Bivariate data should be examined and summarized using the line of reduced major axis (RMA). This line describes the relationship between two correlated variables, but unlike ordinary linear regression, assumes independence of the variables. Whereas the slopes of two RMA's can be statistically compared, there is at present no rigorous method for comparing the positions of two lines (e.g., Jones 1988).
Examples of the use of uni- and bivariate methods to characterize species, distinguish between species, and describe intraspecific and ontogenetic variation include Seitz (1934), Tröger (1986), and Noda (1988). In a few cases, such data have also contributed to studies of evolutionary patterns (e.g., Tanabe, 1973; Noda, 1975).
Shape analysis
With the advent of inexpensive personal computers, alternative methods for the objective quantitative description of morphology have become readily available. In particular, outline-shape analysis is well suited to the study of inoceramids, which generally lack sufficient unique, biologically homologous reference points for landmark analysis (see Temple, 1992 and references therein). Shape analysis provides an objective and conceptually parsimonious complement to qualitative visual processes (Scott, 1980); the eye is particularly adept at identifying differences within small samples, whereas shape analysis can be used to estimate degrees of similarity within large samples.
The different methods of shape analysis fall into three categories: eigenshape analysis, the fitting of polynomial curves, and a family of methods based on Fourier decomposition (e. g., see several papers in Rohlf and Bookstein, 1990). Discussion of their merits has been covered extensively elsewhere (e.g., see Foster and Kaesler 1988, and references therein). Elliptic Fourier analysis (EFA), however, has been favored in several recent studies (e g., Rohlf and Archie, 1984, Ferson et al., 1985, White et al., 1988, Temple, 1992). Ferson et al. (1985) provide a brief introduction to the theory of EFA, and a more detailed account of the methodology and associated problems as applied to inoceramids will be presented in Crampton (in prep.). Unlike many other methods, EFA can describe complex shapes, does not require explicit definition of a biologically homologous or mathematically determined centroid, does not require points on the outline to be equally spaced, and can include simple normalizations for size, position, orientation, and starting position of the trace. A further property of Fourier methods is the ability to invert the transformation and reconstruct an outline from a set of Fourier coefficients. Hence, for example, an "average" shape can be reconstructed from the mean coefficients of a large number of outlines (e.g., Ferson et al., 1985). Elliptic Fourier analysis has been used to demonstrate an association between genotype and morphology within two putative mussel species (Ferson et al., 1985) and to examine patterns of anagenesis and cladogenesis in Albian inoceramids from England (Crampton, 1992, and unpublished data).
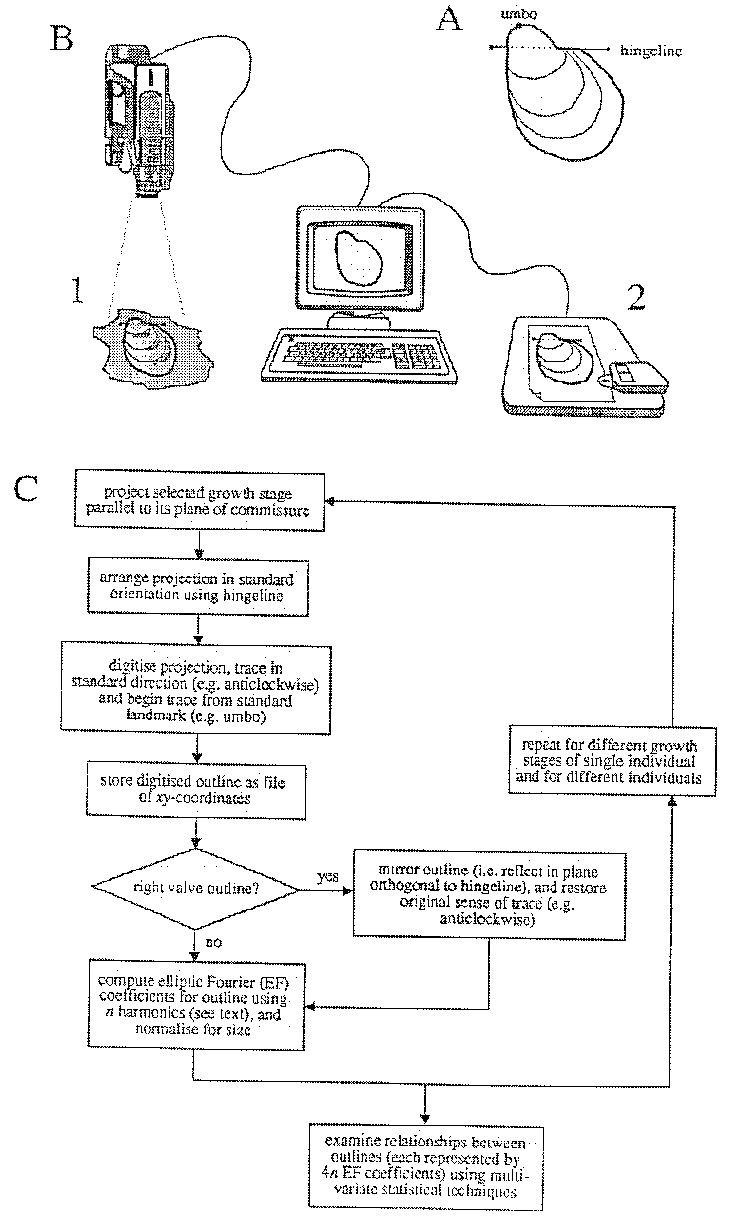
The sequence of steps Crampton's (1992) study are shown in Figure 5 and discussed briefly below. Digitized outlines are generated by manual tracing using either a video camera linked to image-analysis software or a digitizing tablet and photographs or camera lucida drawings (Fig. 5B). Fossil material is generally unsuitable for automated outline capture because of adhering matrix, preservational imperfections, and the desire to trace growth lines other than for the last-preserved growth stage. It is desirable to digitize outlines in a standard fashion (Fig. 5A, C), using the hingeline for orientation, beginning the trace at the umbo, and tracing in a standard direction (e.g., counterclockwise). Standard treatment obviates the need for normalizations during computation of elliptic Fourier (EF) coefficients, normalizations which result in some information loss. Crampton (1992, and unpublished data) has chosen to mirror right valve outlines prior to EFA to remove the effects of primitive bilateral symmetry about the plane of commissure (Fig. 5C). This step permits meaningful comparison of left and right valve shapes, which may be quite different depending on the degree of inequivalvedness. If perfectly equivalved, then a left valve and its mirrored right valve will plot at the same point in multivariate space; separation will increase with increasing inequivalvedness.
The software needed to perform EFA is available with Rohlf and Bookstein (1990) and is written in Fortran for IBM-compatible personal computers. Required input for each outline is a string of xy-coordinates preceded by a sample number and the number of outline coordinates. EFA describes outlines in terms of harmonically-related ellipses, and each ellipse is, in turn, described by four coefficients. Because of the basically elliptical shape of many inoceramids, relatively few harmonics are required to describe their outlines and most of the same information of interest (i.e., the variance) resides in the first three or four harmonics (i.e., first 12 - 16 coefficients, Crampton unpublished data). The number of harmonics required to accurately describe an outline can be estimated in two ways. One can calculate the average discrepancy between the original outline and the inverse Fourier reconstruction based on n harmonics. The Fourier series is truncated at the value of n corresponding to a negligible discrepancy (e.g., smaller than the resolution of hand digitization). Alternatively, one can sum the variance for successive harmonics and compare this sum to the total variance of the Fourier series based upon the maximum possible number of harmonics (equal to half the number of points on the digitized outline). The variance, or power, of each harmonic is equal to half the sum of the squares of the Fourier coefficients. The Fourier series is truncated at the value of n at which, say 99%, of the variance is retained. In shape analysis, the effects of specimen size (which profoundly influences harmonic amplitudes) can be removed during computation of EF coefficients. This normalization utilizes parameters of the first harmonic (i.e., best fitting) ellipse and is probably appropriate in most studies. Information about relative size, however, can be reincorporated into a study during statistical analysis and is essential for an understanding of shape changes through ontogeny.
Elliptic Fourier coefficients for each outline are then treated as variables in a multivariate statistical analysis. The number of outlines should greatly exceed the number of variables per outline. The statistical methods used will vary depending upon the nature of the data and the aims of the study. If there is some a priori knowledge of structure in the data, for example if they can be grouped into stratigraphically or geographically separated populations, then discriminant function or canonical variates analyses might be appropriate. Alternatively, if there is no such knowledge, then cluster or principal components analyses might be used. Where it is used, principal components analysis should probably be based on unstandardized data (i.e., the variance-covariance matrix), rather than standardized data (i.e., the correlation matrix), as is more usual. Although there are theoretical arguments both for and against standardization, it has the disadvantage of giving undue weight (and implied genotypic significance) to high-frequency data and apparently reduces the quality of the analysis (Rohlf and Archie, 1984, Crampton unpublished data).
In summary, biometric methods are becoming increasingly important in the study of inoceramid taxonomy and paleobiology. In particular, outline shape is a fundamental aspect of morphology which is suited to biometric description, using either linear and angular measurements, or Fourier shape analysis. Using such methods, it is possible both to quantify and to objectively compare ontogenetic and intra- and interpopulation variations in morphology. This information is crucial to the interpretation of apparently complex patterns of morphological change through space and time.
LOWER TURONIAN GENERA OF INOCERAMIDAE
The genera and subgenera of Inoceramidae are critically in need of revision. The proliferation of generic names by Heinz (1932), some of them nomina nuda or nomina dubia (Cox, 1969), created chaos in inoceramid taxonomy. The names not only were improperly formulated, lacking generic diagnoses and, in some cases, proper designation of type species, but they also were based solely upon subtle differences in external shell form and sculpture. The nature of phenotypic and ecophenotypic population variation, parallel evolution, and homeomorphy in external shell features was not considered by Heinz (1932) or the majority of early inoceramid workers (Kauffman and Powell, 1977). Most inoceramid specialists now recognize that homeomorphy in shell shape and external morphology is a common phenomenon, not only within the Inoceramidae but also between this family and related Permian inoceramid ancestors such as Atomodesma, Kolymia, Intomodesma, and Aphanaia (all Ambonychiidae; Kauffman and Runnegar, 1975). Parallel and convergent evolution in shell form compounds the difficulties of establishing a comprehensive taxonomy for the group.
Cox (1969), therefore, took a conservative view of generic and subgeneric classification of the Inoceramidae in the Treatise on Invertebrate Paleontology. He placed the great majority of Heinz's (1932) genera into synonymy (mainly with Inoceramus s.s.) and utilized as subgenera (formally authored by Cox) only the new names of Heinz for which at least a skeletal description of generic characteristics had been presented or those which had been subsequently used in published literature (e.g., Cataceramus, Cremnoceramus and Spyridoceramus). The majority of inoceramid workers have followed Cox's lead. Annie Dhondt (personal communication, Dec. 6, 1993), however, has pointed out that, according to the ICZN rules of zoological nomenclature, all of Heinz's (1932) new genera to which he had assigned a valid, previously described and illustrated species as the genotype, are still valid genera, whether or not they were ever formally described. This may invalidate many of Cox's (1969) assignments of Heinz's (1932) genera to "nomina nuda.". Unfortunately, Cox died before these mistakes could be rectified in the Treatise on Invertebrate Paleontology (1969). In future revisions of inoceramid taxonomy, retention of Heinz's (1932) generic names must be carefully considered on a case-by-case basis.
A few names used by Cox (1969) as subgenera have subsequently been formally elevated to generic status (e.g., Mytiloides; Kauffman and Powell, 1977) or used as genera in non-systematic faunal analyses (e.g., see Kauffman, 1975, 1976a,b,c; Kauffman et al., 1976, 1993 in press). Further, several of Heinz's (1932) names considered by Cox (1969) as nomina nuda, were subsequently formally described as subgenera (e.g., Platyceramus, Endocostea, Magadiceramus, Cordiceramus) by Seitz (1961, 1965, 1967). But our modern knowledge of the Inoceramidae, based on a broader suite of external and internal shell features, suggests that the elevation of several subgeneric names to generic status, and the formulation of additional genera and subgenera within the family is warranted.
Kauffman (1994, in manuscript) is currently revising the genera and subgenera of the Inoceramidae utilizing, for the first time, a combination of external shell form and ornament (including ontogenetic changes), with internal morphological characteristics such as the shape, kinds and position of muscle insertion areas, the nature and position of the pallial line relative to the commissure, characteristics of the ligament and ligamental plate, the umbonal septum, pseudodentition, and the nature of the byssal slit, where present. Data on these characters are commonly difficult to obtain, especially for the weakly impressed muscles. Internal shell characteristics appear to be genetically conservative and clearly divide the Inoceramidae into natural morphologic groups. A combination of internal and external shell features best characterize natural generic and subgeneric divisions of the Inoceramidae, and may eventually allow phylogenetic details to be worked out in great detail. In many cases, generic and subgeneric divisions of the Inoceramidae based on internal shell characters separate taxa which were formally grouped under a single genus or subgenus based solely on external shell features, thus defining parallel evolutionary trends.
At present, four inoceramid genera are commonly recognized in the Early Turonian of the Europe and the Americas (others may exist among rare and geographically isolated groups). These are: Inoceramus s.s. J. Sowerby; Mytiloides Brongniart; Sergipia Maury; and Cladoceramus Heinz (1932) (?=Rhyssomytiloides Hessel, 1988; see below). A diagnosis and description of each of these genera is presented below, reflecting the concepts generally agreed upon in the 1992 Hamburg meeting (dissenting views are cited for each genus, where relevant). The genus Cremnoceramus was also discussed at this workshop, and its possible relationship to geniculate specimens of Late Turonian Mytiloides? incertus (Jimbo) (= M. fiegei fiegei (Tröger) ). But inasmuch as no Lower Turonian Cremnoceramus are yet recorded, redescription of this genus is not included herein. (This section was initially prepared by E. G. Kauffman with descriptions taken from Kauffman and Powell, 1977, as modified herein; Kauffman, 1994, generic revision in manuscript; Kauffman and Villamil, 1994, in manuscript for generic treatment of Sergipia ; the current version incorporates all comments and revisions subsequently offered by workshop participants).
GENUS INOCERAMUS J. Sowerby, 1814
TYPE SPECIES: Inoceramus cuvieri Sowerby (Cox, 1969, p. N315 by subsequent designation).
DIAGNOSIS: Adult shells attaining >1 m height. Inequivalve, left valve largest, most inflated; prosocline, outline erect-ovate, subquadrate, or subtrapezoidal; projecting beaks, umbones prosogyrous to orthogyrous. Adult ornament of concentric rugae with intercalated growth lines or lamellae; juvenile ornament distinct. Well-defined posterior auricle, auricular sulcus; post umbonal sulci, folds common. Flattened anterior face with sinuous, slightly gaping byssal slit. Moderately thick prismatonacreous shell; calcite prisms elongated, needle-like. Ligamental plate thick, elongated, bearing numerous close-set, subrectangular, vertically elongated resilifers; umbonal septum and subumbonal cavity prominent, subtriangular. Musculature weakly impressed; pallial line thin, continuous or anteriorly pitted, at distal edge of small radial muscle tracks; posterior adductor boat-shaped, submarginal; large pedal-byssal retractor muscle insertion area below umbonal septum; 1-3 small, ovate, dorsoanterior pedal-byssal protractor insertion areas situated just outside of pallial line.
GENERIC DESCRIPTION: Small to large size (>1 m) adult shells; slightly to strongly inequivalve with left valve largest and most inflated. Shells prosocline; most commonly with ovate, subquadrate, or subtrapezoidal outline; beaks, umbones inflated, prosogyrous to orthogyrous, normally projecting above hinge line and anteriorly situated. Ornamentation characteristically of weakly to strongly developed, unequally to subequally spaced, concentric rugae with numerous, closely spaced, evenly to subevenly developed growth lamellae or raised growth lines between rugae. Many species with fine, discrete, juvenile ornamentation on umbo, consisting of closely and evenly to subevenly spaced raised concentric growth lines without rugae. Posterior auricle well-defined, triangular, separated from disc by auricular sulcus; a post-umbonal sulcus occurs in some species, as do very small anterior auricles ("ears"). Byssal slit sinuous, slightly gaping in many species, bounded by a flattened anterior face or a pseudolunule with low marginal ridges. Small, irregular, sinuous costellae may occur on anterior face or in pseudolunule at near-right angles to the growth lines. Shell moderately to very thick, especially prismatic layer, with greatest thickness along the broad posterior ligamental plate; resilifers numerous, closely spaced, narrow and vertically oriented, shallow to moderately excavated, separated by narrow, rounded to flattened ridges. Umbonal septum subtriangular, moderate to large size, with prominent subumbonal cavity. Some species exhibit a poorly defined pseudocardinal "tooth" (an inward extension of a small anterior auricle or the thickened dorsoanterior shell margin), with or without a shallow socket on the opposite valve at the anterior end of the umbonal septum. Muscle attachment areas weakly impressed. Pallial line thin, continuous or mostly so, situated just inside the commissure; some species have a pitted pallial line segment dorsoanteriorly; mantle attachment/retractor muscles, which form pallial line, leave radial tracks (shallow faint grooves) on shell interior except in the posterior adductor track. Some accessory mantle attachment/retractor muscles occur outside pallial line on inner surface of posterior auricle. Monomyarian; posterior adductor muscle insertion area faint, situated near posteroventral margin, typically elongated and boat-shaped. A large, well impressed, pedal-byssal retractor muscle attachment area occurs on the underside of the umbonal septum in many species (unknown in others); one to three small, ovate to vertically elongate-ovate pedal-byssal protractor muscle insertion areas occur dorsoanteriorly, just outside of the pallial line, in numerous species. Gill suspender and other muscles unknown to date. Prodissoconch known on a few species, large and inflated, indicating long-lived planktotrophic larvae.
DISCUSSION: Kauffman and Powell (1977) utilized Inoceramus as a subgenus within the genus Inoceramus J. Sowerby to identify a lineage of Cenomanian-Turonian taxa that was characterized by erect, flattened to moderately inflated forms with weakly to moderately developed rugae, subequal growth lines or lamellae, and which lacked strong posterior and anterior sulci and folds. Thus defined, Inoceramus (Inoceramus) includes Cenomanian Euramerican species such as I. (I.) pictus Sowerby and subspecies, I. (I.) prefragilis Stephenson, I. (I.) virgatus Schlüter and subspecies, I. (I.) scalprum Woods, some illustrated versions of I. tenuis Mantell (although the type of I. tenuis may belong to Birostrina), and closely related species and subspecies of these taxa. Inoceramus (Inoceramus) also includes Lower and Middle Turonian I. (I.) cuvieri Sowerby (the type species of Inoceramus), I. (I.) apicalis Woods, I. (I.) inaequivalvis Schlüter, I. (I.) saxonicus Petrascheck, I. (I.) tuberosus Keller, and closely related species and subspecies. Kauffman and Powell (1977, p. 71) suggested that the Cenomanian lineage of Inoceramus arvanus Stephenson - I. rutherfordi Warren, characterized by strong posterior folds and sulci, and less commonly, anterior sulci on the shell exterior, should be placed in a different subgenus (unnamed) of Inoceramus. We further suggest that the strongly sulcate, coarsely rugate, posteriorly auriculate species plexus centered around Inoceramus lamarcki might be treated similarly. Inoceramus s.s. is not known below the Cenomanian and ranges at least into the Middle Coniacian, but forms provisionally assigned to Inoceramus s.s. are reported in rocks as young as Late Campanian and Early Maastrichtian (Kauffman, et al., 1993, in press). Many pre-Cenomanian and post-Coniacian species assigned to Inoceramus can now be placed in other genera; some are homeomorphic on Inoceramus s.s. and can be differentiated mainly on interior shell features, musculature, and ligamenture (Kauffman, 1994, in manuscript).
GENUS MYTILOIDES BRONGNIART, 1822
TYPE SPECIES: Ostracites labiatus Schlotheim (by monotypy) = Inoceramus (Mytiloides) labiatus (Schlotheim) fide Cox, 1969, p. N320; = Mytiloides labiatus (Schlotheim) fide Kauffman and Powell, 1977, p. 71-72.
DIAGNOSIS: Moderate size, length <50 cm; slightly to moderately inequivalve; left valve largest, more inflated. Shape subcircular to characteristically labiatoid; prosocline. Beaks, umbones inflated, slightly-moderately projecting, prosogyrous to nearly orthogyrous; ventral umbo commonly geniculated. Characteristic ornament of weakly to strongly developed rugae intercalated with equally to unequally developed growth lines and lamellae. Juvenile ornamentation distinct. Posterior auricle flat, triangular, separated from disc by moderately excavated to broad, shallow auricular sulcus. Anterior face rounded to subtruncated; byssal slit slightly sinuous, poorly defined to absent. Ligamental plate posterior, short to moderately long, thin, delicate, bearing small, moderately spaced, slightly excavated, subtriangular to ovate resilifers separated by low, flat to rounded platforms. Umbonal septum small or absent. Muscles very weakly impressed; pallial line continuous, submarginal, without radial muscle tracks; posterior adductor insertion area submarginal, posteroventral, narrow, elongated; no pedal-byssal muscle insertions known. Shell prismatonacreous, very thin, with short, blocky, calcite prisms.
GENERIC DESCRIPTION: Shell of moderate size, rarely exceeding 30 cm in length, ranging to 50 cm; subequivalve to moderately inequivalve with left valve slightly larger and more inflated; shell slightly to moderately biconvex. Valves moderately to highly prosocline; beaks, umbo anteriorly situated except in M. latus group (beaks just anterior to midline), strongly prosogyrous to nearly orthogyrous, slightly to strongly projecting above the hinge line. Low geniculation occurs at ventral edge of umbo, between juvenile and adult portions of the disc, on many species. Shell outline ovate, subovate, elongate-ovate, or tongue-shaped (labiatoid) on various species. External ornamentation characterized by equally to unequally spaced, weakly to strongly developed concentric rugae with regularly (typical) to irregularly developed raised growth lines and/or flat lamellae between rugae over most or all of the valve surface. Many species lack growth lines in the juvenile and/or early adult growth stages; rare species lack them altogether, or lack rugae over most of shell. Juvenile ornamentation commonly distinct from adult, consisting of closely spaced, subequally developed, fine to coarse, raised growth lines without rugae. Rare species (e.g., undescribed smooth form) have small anterior projection in front of beak ("ear" or anterior auricle). Posterior auricle small to moderate size, flattened, subtriangular, in some cases flared posteriorly forming an acute auricular angle; posterior auricular sulcus poorly defined as a broad shallow depression between auricle and disc, accentuated in some species where umbonal area is highly inflated. Anterior face normally curved, convex, with a poorly defined, slightly sinuous byssal slit or none at all; a few species have a flattened to slightly concave anterior face and a more prominent, slightly gaping, sinuous byssal slit. Shallow anterior radial sulcus occurs on rare species (e.g., M. submytiloides). Hinge line short to moderate length, extending posterior to beak, umbo; ligamental plate very thin, delicate, bearing small, subtriangular to ovate, slightly excavated resilifers separated by broad low ridges or narrow flattened platforms. Umbonal septum very small or absent; subumbonal cavity not significantly excavated. Musculature very weakly impressed. Pallial line continuous (entire), situated just inside the commissure, normally lacking radial traces of mantle attachment muscles. Narrow, laterally elongated, posterior adductor insertion area situated posteroventrally near to the commissure. No traces of pedal-byssal or gill suspender muscles observed. Shell very thin (typically ranging from 0.1-0.3 mm), especially the calcite prismatic layer, relative to other Inoceramidae; prisms small and blocky.
DISCUSSION: Brongniart (1822) originally described Mytiloides as a genus, but without a comprehensive diagnosis. Until recently, most authors have placed the name in synonymy with Inoceramus (e.g,. Seitz, 1934, 1965). Cox (1969) utilized Mytiloides as a subgenus of Inoceramus. Kauffman and Powell (1977) re-described Mytiloides Brongniart as a full genus, noting that it differed from typical Inoceramus in its strongly prosocline form, low shell inflation, subequivalve shells lacking strong radial sulci and folds, very thin nacreous and prismatic shell layers (especially in the hinge and umbonal areas), and weak byssal slit, or none at all. Internally, Mytiloides is further distinguished from Inoceramus by the lack of preserved pedal-byssal muscle insertion areas; by its thin continuous pallial line without impressed, radial, mantle retractor muscle tracks; by an elongated, subcrescentic, posterior adductor insertion area and a thin, narrow, ligamental plate with small, subtriangular, weakly impressed resilifers; and by its small umbonal septum, seemingly without a deeply excavated subumbonal cavity. Species of Mytiloides can be divided into at least two major morphologic groups of potential subgeneric rank: (a) Mytiloides s.s. consisting of mytiloid-shaped, strongly prosocline taxa (e.g., M. mytiloides, M. labiatus, M. striatoconcentricus lineages), including the oldest (Late Cenomanian) Mytiloides known (M. n. sp.; smooth form, M. submytiloides, M. hattini); and (b) rounded to ovate, suberect Mytiloides such as M. "latus" (sensu Woods, 1912, Fig. 41; other illustrated types of Woods are probably Inoceramus waltersdorfensis), and possibly M. incertus (Jimbo) (Note that some participants at the Workshop would place this species in Cremnoceramus because it has an adult geniculation; we have retained it here in Mytiloides because adult geniculation is also common in more inflated members of this genus, and M. incertus is otherwise similar to more rounded Mytiloides of the Early Turonian). This rounded clade of Mytiloides will receive a new subgeneric name in Kauffman's forthcoming inoceramid revision (in manuscript, 1994). Mytiloides s.s. originates in the Late Cenomanian and ranges through the Turonian, Coniacian, and possibly Early Santonian, although its record is not continuous. Older Mytiloides-like species reflect homeomorphy and belong to different genera, as indicated by interior shell features, musculature, and ligamenture (Kauffman, 1994, in manuscript).
GENUS SERGIPIA MAURY, 1925
TYPE SPECIES: Inoceramus (Sergipia) posidonomyaformis Maury, 1925; = Sergipia posidonomyaformis (Maury) fide Cox, 1969
DIAGNOSIS: Moderately small adult shells, length <15 cm, slightly inflated, subequivalve, left valve slightly larger than right. Shape subrounded to ovate; length > height, rounded growth-line trace; slightly prosocline. Beaks subcentral, very slightly projecting, slightly prosogyrous to orthogyrous. Anterior, posterior auricles subtriangular, poorly defined from disc by broad concave trough. Surface ornament of subequally spaced low rugae and/or faint to strongly raised, close-set, growth lines and/or lamellae. Ligamental plate very thin, delicate, extending on both sides of beak, bearing moderately to widely spaced, weakly excavated, subtriangular to ovate resilifers. No umbonal septum. Short, prominent internal rib separates posterior auricle from disc. Musculature very weakly impressed; poorly known; pallial line complete, very thin,with faint radial muscle tracks; posterior adductor insertion area small, ovate, posteroventral near commissure; no pedal-byssal muscles or byssal slit known. Shell prismatonacreous, exceptionally thin, with short blocky calcite prisms.
GENERIC DESCRIPTION: Shell attaining moderate size; length rarely exceeding 10 cm; adult shells averaging 4-5 cm in length. Shell slightly inflated to flattened, with greatest inflation dorsocentrally at base of umbo. Subequivalve, with left valve very slightly larger than right valve. Shape subrounded, ovate, rarely elongate-ovate with length greater than height; anterior, ventral, and posterior margins with rounded trace. Slightly prosocline; beak and umbo slightly prosogyrous to orthogyrous, beak subcentral to slightly anterior of midline, only slightly projecting above long dorsal margin, which extends both anterior and posterior to beaks. Anterior and posterior auricles triangular to subtriangular, with angular to somewhat rounded dorsolateral margins; auricles either merging continuously with disc or separated from it by very shallow broad auricular troughs. Surface sculpture consisting of subequal, closely spaced, small rounded rugae or large raised growth lines on umbo; rugae become separated by a few, equally to unequally distributed, fine raised growth lines on the adult disc; growth lines more prominent than rugae on some species. Internally, ligamental plate very thin, rounded, extending both anterior and posterior to beak (anterior ligament plate shorter), bearing small, moderately to widely spaced, subtriangular, weakly to very weakly excavated resilifers on both sides of beak. Surface sculpture also moderately defined on interior of very thin prismatonacreous shell characterized by short blocky prisms. A prominent, narrow, rounded internal rib extends from the posterior umbo to or near the commissure at the junction between the posterior auricle and the disc. No umbonal septum known. Musculature very weakly impressed and poorly known. Posterior adductor insertion area small, ovate to subrounded, located near the ventroposterior commissure. Posterior adductor muscle track defined by small, weak lateral grooves on shell interior. Faint, incomplete traces of very small radial grooves suggest mantle attachment muscle tracks; pallial line entire where observed, but incompletely known. No pedal-byssal or gill suspender muscles observed; byssal slit, if present, not differentiated on observed specimens; species may not be byssate as adults.
DISCUSSION: The extension of the resilifer-bearing ligamental plate anterior to the beaks places the assignment of Sergipia to the Inoceramidae (Cox, 1969) in question. The same might be said for Inoceramya Ulrich, which is inferred to have had similar distribution of resilifers (Cox, 1969, p. N317, fig. C47-5b). An alternative placement might be in the Posidoniidae, which are externally very similar in form and general ornamental characteristics, and which have the ligamental plate extended anterior to the beaks in most species. But the presence of multivincular resilifers along the anterior and posterior hinge plate of Sergipia, features not yet known from the Posidoniidae, supports placement within the Inoceramidae at this time. This taxonomic assignment is also supported by the external similarity in form and ornamentation of Sergipia to the inoceramid genus Steinmannia, which also has the ligamental plate extended anterior to the beaks, but has resilifers restricted to its posterior segment (Cox, 1969, fig. C49-3b), and to the Mytiloides "latus " (sensu Woods, 1912, Fig. 41 only) species group. In fact, some authors (e.g., Hessel, 1988) have assigned species that apparently belong to the M. "latus " lineage, and which lack anterior extension of the ligamental plate, to Sergipia (e.g., S. hartti Hessel), and vice versa, based solely on the similarity of external shell form and ornamentation. We suggest that Sergipia may have been derived from rounded Lower Turonian M. "latus" (sensu Woods, 1912, Fig. 41) by changes in the ligamental plate, normally a conservative character within inoceramid genera and subgenera. Kauffman and Villamil (1994, in manuscript) are currently undertaking a taxonomic revision and documenting the early evolution of Sergipia from the American species.
GENUS CLADOCERAMUS Heinz, 1932
TYPE SPECIES: Inoceramus undulatoplicatus var. michaeli Heinz, 1928, p. 76 (=Inoceramus digitatus Schlüter (non Sowerby) 1877, p. 267, pl. 36; fide Seitz, 1961, p. 95).
DIAGNOSIS: Small (Turonian) to very large (Santonian; >1 m axial length) adult shells; subequivalve to slightly inequivalve; left valve slightly larger. Outline ovate to elongate-ovate to subtrapezoidal; moderately prosocline. Beaks, umbones prosogyrous, anterior, slightly projecting; umbo non-geniculate. Anterior face rounded; no byssal slit observed. Posterior auricle relatively small, subtriangular; auricular sulcus slightly to moderately concave. Juvenile umbonal ornament of fine to coarse raised growth lines or small rugae; adult ornament characteristically of weakly to strongly developed, divaricate radial plicae or folds, most strongly developed but fewer in number on the posterior flank of the disc, but in some cases only developed anteriorly. Relatively weaker concentric ornament of coarse raised growth lines and small rugae. Shell prismatonacreous, moderately thin. Ligamental plate relatively thin for shell size, bearing numerous, closely spaced, small, slightly-moderately excavated resilifers. Muscles weakly impressed. Posterior adductor insertion area large, canoe-shaped, close to posteroventral margin; pallial line thin, continuous, incompletely known; no pedal-byssal muscle insertion areas known. Umbonal septum small, subtriangular.
GENERIC DESCRIPTION: Adult shell small (Early Turonian) to very large size (>1 m in Early Santonian); subequivalve to slightly inequivalve with left valve slightly larger and more convex dorsally; shell slightly to moderately biconvex, moderately prosocline. Shell outline ovate, subovate, elongate-ovate to subtrapezoidal. Beaks, umbones moderately prosogyrous to suberect, situated at or near anterior end of a short to moderately long hinge line; beaks slightly projecting dorsally above the hinge axis. Anterior margin straight to slightly rounded (convex outward), bending slightly to moderately inward to commissure, but without strong anterior truncation or development of a pseudolunule. No byssal slit observed, but anterior face poorly known. Posterior auricle small relative to shell size, subtriangular, weakly to moderately defined, separated from disc by slightly to moderately concave auricular sulcus on dorsoposterior flank of umbo, or the flank of the first divaricating fold on the disc. Lateral and ventral margins of adult shells slightly to moderately curved, with undulating commissure at intersection of radial folds or plicae. Surface of juvenile shell on umbo characterized by a predominance of concentric ornamentation (fine to coarse raised growth lines or small rugae; in some cases nearly smooth or with weakly developed radial costae); adult disc characterized by small to large divaricate radial plicae or folds which extend ventrally and curve laterally from a median line or discontinuous ridge approximating the growth axis of the shell; posterior plicae or folds commonly fewer and larger than those anteriorly; individual plicae and folds may bifurcate distally. Some species have plicae, folds weakly developed on one flank (usually the anterior) of the shell. Coarse, moderately to broadly spaced, raised, subequal to unequal concentric growth lines and/or small rugae cross folds, plicae; raised growth lines commonly become denser and/or coarser and more evenly spaced on umbo, with or without radial elements. Shell moderately thin, becoming thicker near ligamental plate; plate relatively thin for size of shell, bearing small closely spaced, slightly to moderately excavated resilifers. Posterior adductor insertion area large, canoe-shaped, situated moderately close to the posteroventral commissural margin. Some species have small umbonal septum. Pallial line partially known, thin and entire. No other internal features are well known at present.
DISCUSSION: Among the genera of Inoceramidae discussed at the workshop, Cladoceramus was the focus of greatest debate. Heinz (1932) proposed the new genus Cladoceramus and designated Inoceramus michaeli (= I. undulatoplicatus var. michaeli Heinz) as the type species. Heinz (1932) synonymized "I. digitatus" Schluter, 1877 (non I. digitatus Sowerby, 1829) with I. undulatoplicatus michaeli Heinz (1928) in designating the genotype species. Heinz (1932, p. 25) never described or illustrated his new genus, as noted by Cox (1969; p. N320), who stated that Cladoceramus was a nomen nudum and synonymized this genus with Sphenoceramus J. Böhm 1915 (type species Inoceramus cardissoides Goldfuss, 1836; subsequent designation by Vialov, 1960) in the Treatise on Invertebrate Paleontology. However, Cox (1969) did not take in to account the ICZN ruling that a genus does not have to be described to be valid if the genotype species is validly described at the time the new genus is proposed (Annie Dhondt, personal communication, 1993) and further overlooked the work of Seitz (1961, 1965), who noted many morphological distinctions between Cladoceramus, as represented by Inoceramus undulatoplicatus and related species, and Sphenoceramus, as represented by I. cardissoides. Unfortunately, Cox died before these oversights could be corrected in his 1969 Treatise article. Seitz (1961) further validated the use of this name by providing a diagnosis of Cladoceramus (as a subgenus of Inoceramus), and illustrated typical species, i.e., I. (C.) undulatoplicatus Roemer, and I. (C.) japonicus Nagao and Matsumoto (1961, 1965). Seitz noted that: (a) Heinz's genus Cladoceramus should have the rank of subgenus; (b) that Woods (1912) had clearly separated I. digitatus Schlüter (a Cladoceramus) from I. digitatus Sowerby (a Sphenoceramus?); (c) that I. digitatus Schlüter, originally described as a subspecies, was elevated to species rank (Heinz, 1932); (d) that Cladoceramus was most closely related to the subgenus Platyceramus, some species of which had weakly developed flared plicae and folds (e.g., I. (P.) cycloides wegneri; I. (P.) rhomboides heinei). Cladoceramus was separated from Platyceramus by placing all species with divaricating radial plicae or folds, which were stronger than the concentric ornament, into Cladoceramus (the concept basically used today); and (e) that Cladoceramus was not closely related to, nor evolutionarily transitional with, the genus Sphenoceramus as represented by S. pachti, S. cardissoides, S. steenstrupi and S. schmidti. Seitz (1965) subsequently defined and illustrated the subgenus Sphenoceramus in depth; Cox (1969) re-elevated Sphenoceramus to generic rank. In his 1961 paper, Seitz correctly attributed the subgenus Cladoceramus to Heinz (1932), even though he proposed this name without diagnosis. Kauffman (1975, 1991) and Kauffman et al. (1993, in press) has subsequently used the name Cladoceramus as a full genus, but without formal justification; this is presented below.
Subsequent work on inoceramid genera confirms Seitz's observations that Cladoceramus and Sphenoceramus are morphologically quite distinct, and only distantly related. Sphenoceramus is acutely triangular in shape, with a strongly projecting beak and umbo, a short hinge line and very small posterior auricle. Various species either lack radial ornamentation or have radial ribs weakly to moderately developed, but never bifurcating, divaricating, or stronger than the concentric ornamentation on the disc. Further, Sphenoceramus has a very well defined posterior umbonal folds and sulci (especially the deep posterior auricular sulcus) and may have weakly defined anterior sulci on the disc. Sphenoceramus has a diagnostic concentric ornamentation of very large, asymmetrical, subevenly developed, angular to subangular, concentric rugae between which are found a few coarse raised growth lines which become more regularly developed dorsally, and weaker ventrally. Internal characteristics also seem to be very different between these genera, although they are not yet fully known. Sphenoceramus has a much thicker ligamental plate, relatively larger resilifers, and a much larger, more quadrate to ovate posterior adductor muscle insertion area than Cladoceramus. Mantle retractor muscle tracks are common and may be well-defined on the interior shell surface. Sphenoceramus also has a truncated anterior face, in some cases a lanceolate pseudolunule, and a long sinuous byssal slit not found on Cladoceramus. In Kauffman's revision (1994, in manuscript), Cladoceramus and Sphenoceramus are regarded as discrete genera of the Inoceramidae based on a survey of both internal and external shell features. Both differ significantly from Inoceramus in shape, size, ornamentation, development of their folds and sulci, musculature, and the possession of some prevalent form of strong radial surface sculpture. Cladoceramus is typical of the Early Santonian of Europe and North America (Seitz, 1961; Kauffman, 1975; Kauffman et al., in press), but derived species range throughout the Santonian.
It now appears, however, that Cladoceramus had its origins in the Early Turonian of Brazil, in the ammonite zone characterized by Mammites, Kamerunoceras, Neoptychites and Watinoceras spp. (ammonite zone 2 of Bengtson, 1983). Hessel (1988) described five species of a new genus, Rhyssomytiloides, in this middle Early Turonian ammonite zone from the Sergipe Basin, Brazil: R. mauryae, R. bengtsoni, R. alatus, R. beurleni, and R. retirensis. These species have external characteristics identical to those of Cladoceramus, including: (a) coarse divaricating radial folds or plicae on the adult portion of the shell, which are stronger than the concentric ornamentation; (b) strongly developed, raised, subequal growth lines or small rugae on the umbo and early adult portions of the disc; (c) an undulating commissure where the folds intersect it; (d) a moderately prosocline shell with a slightly curved growth axis; (e) a small, subacute, subanteriorly situated beak which barely projects above the hinge line; and (f) a somewhat flared, slightly undulating, dorsoposterior auricle which is poorly differentiated from the disc by a broad sulcus. In fact, Hessel's species are very similar to the earliest growth stages of large I. (Cladoceramus) undulatoplicatus undulatoplicatus, and I. (C.) undulatoplicatus michaeli illustrated by Seitz (1961, pl. 5,6), suggesting that evolution of the group between the Early Turonian and Early Santonian mainly involved great expansion and modification of the adult growth stage. Inasmuch as interior shell characters of the species placed by Hessel (1988) in Rhyssomytiloides are still poorly known (a large posterior adductor muscle insertion area, and small resilifers along the relatively thin ligamental plate near the beak), parallel evolution of discrete inoceramid genera to produce this shell form cannot be demonstrated. Thus, at this time Rhyssomytiloides Hessel is best placed in synonymy with Cladoceramus Heinz (1932). Whereas Hessel (1988) discussed differences between Rhyssomytiloides and Sphenoceramus Böhm, to which she had originally assigned R. alatus and R. mauryae, no generic level comparisons were made to Cladoceramus. Hessel did note, (1988, p. 28), however, very close similarities between R. mauryae and two common Cladoceramus species, "Inoceramus" undulatoplicatus Roemer, and "I. (Platyceramus)" japonicus japonicus. She distinguished R. mauryae from these species primarily on shell size, thickness, inclination, modest differences in inflation, and varying development of the concentric ornamentation. None of these are deemed generic-level distinctions. The validity of Rhyssomytiloides as a discrete inoceramid genus will depend upon the results of detailed study of the internal shell features and their comparison to those of Cladoceramus.
At the Hamburg inoceramid workshop, debate regarding Cladoceramus as a valid genus in the Early Turonian focused on two things: (1) whether or not Cladoceramus should be placed in synonymy with the genus Platyceramus , and (2) whether or not Rhyssomytiloides should be allowed to stand as a genus, rather than being synonymized with Cladoceramus.
Specifically, Matsumoto, Noda, and Kozai (1982), Noda (1983), and Lopez (1986, 1992, personal communication, 1993), among others, have recommended placing Cladoceramus in synonymy with Platyceramus, which has page priority over Cladoceramus , and have noted transitional forms between them, even within single populations (e.g., Platyceramus higoensis; Noda, 1983). Collectively, these authors have pointed out similarities in shape, convexity, and concentric ornamentation between Cladoceramus and Platyceramus. The only difference between them, as currently defined, is that radial and divaricating ornamentation is stronger than the concentric ornamentation in species assigned to Cladoceramus, whereas some Platyceramus species have poorly developed radial ornamentation that is more weakly defined than the concentric ornamentation, and which occurs more irregularly in populations. Such transitional forms are not yet known, however, in the genotype species of Platyceramus. Annie Dhondt also now favors this view (personal communication, Dec. 5, 1993), in contrast to her generic usage of Cladoceramus in Dhondt and Dieni (1990). Seitz (1961), in contrast, favored retaining Cladoceramus as a subgenus for those inoceramids with divaricating radial ornamentation that is more prominent than the concentric ornamentation, although he clearly noted difficulties in assigning a genus to transitional species. Kauffman (1975, 1991) and Kauffman et al. (1993, in press) have used Cladoceramus as a full genus, and placed within it all large Santonian inoceramids in this plexus with radial or divaricating ornamentation of any sort within species populations, including some taxa formally placed within Platyceramus. Both views have validity and need to be further explored until ontogenetic development, internal shell characteristics, and ornamental development of the shells of Platyceramus and Cladoceramus spp. can be critically compared; this is beyond the scope of the present paper and the principal authors choose to retain Cladoceramus as a valid genus until this careful systematic work is completed.
Finally, three workshop participants (Tröger, Lopez, Hilbrecht) expressed the view that Rhyssomytiloides Hessel should not be placed into synonymy with Cladoceramus until interior shell structures can be compared to prove the linkage. This view, however, promotes the continued use of a newly proposed genus that cannot be differentiated in any substantial way from a previously described genus with priority. Rhyssomytiloides, except for its relatively small size, appears to have identical ontogenetic development, shell shape and ornamentation to species now placed within Cladoceramus (e.g. C. undulatoplicatus), and because maximum adult shell size of a population from a single region is rarely even regarded as a species-level characteristic within clades because of potential ecological controls, there is no justification for retaining Rhyssomytiloides as a distinct genus. There are no known characters to separate it from Cladoceramus spp. at present (or from Platyceramus, if Cladoceramus is considered a synonym of this genus). The fact that a time gap exists between the Turonian and Santonian occurrences of Cladoceramus is not justification for generic separation. Many inoceramid lineages show these gaps (e.g., Early and uppermost Turonian occurrences of Mytiloides, and Permian and Jurassic occurrences of the Family Inoceramidae, without intermediate representatives).
LOWER TURONIAN INOCERAMID BIOSTRATIGRAPHY
The Inoceramidae are exceptionally good biostratigraphic tools among Bivalvia because of an unusual combination of traits (Kauffman, 1975). The great majority of known inoceramid species have intercontinental to cosmopolitan distribution in normal marine, temperate zone facies; they are much less common, but still widespread, in the Tethyan Realm. When compared to ammonite and plankton zonal boundaries or to widespread event surfaces/intervals, the Inoceramidae appear to have had rapid, widespread dispersal mechanisms; their range boundaries commonly approximate (but do not equal) chronostratigraphic surfaces. Dispersal was apparently by very long-lived planktotrophic larvae, as is also suggested by large inflated prodissoconchs known from a few species. Species durations of biostratigraphically useful Inoceramidae are remarkably short for such a cosmopolitan group, ranging from 0.12 - 0.5 Ma per range zone in middle and Upper Cretaceous strata of the Western Interior Basin of North America (Kauffman, 1975; Kauffman et al., 1993, in press), where they can be compared to new, closely spaced,single crystal 40Ar-39Ar radiometric ages (Obradovich, 1993, in press). They evolved at rates comparable to those of ammonites and much faster than those of marine plankton in the Cretaceous. Normally, broad biogeographic dispersal of large populations slows evolutionary rates in marine organisms (e.g., Jablonski, 1986), so that the Inoceramidae are unusual in this respect. Finally, the Inoceramidae are the numerically dominant macrofossil in most Cretaceous fine-grained facies, especially those representing oxygen-restricted benthic environments (e.g., Sageman, 1989, Kauffman and Sageman, 1990), and are an important component of communities in coarser-grained nearshore facies as well. Their resistant, organically bound prismatic calcite shell layer preserves in most marine facies, including those where ammonites may be partially or wholly dissolved during early diagenesis. These characters enhance the biostratigraphic utility of the Inoceramidae in Jurassic and Cretaceous sequences worldwide.
Several biozonal schemes have been proposed for Lower Turonian Inoceramidae. Initially, authors in both Europe and America utilized a single zone, that of "Inoceramus labiatus" (s.l.) for this substage (e.g., Cobban and Reeside, 1952). Kauffman (1975, 1976 a,b,c), Kauffman et al. (1976, 1993, in press), Seitz (1934), Tröger (1981), and Walaszczyk (1992), among others, have proposed a more refined lineage zonation based on rapidly evolving Lower Turonian species within the genus Mytiloides. Initially, following the systematic concepts of Seitz (1934), this zonation consisted of (in ascending order) the zones of M. submytiloides, M. opalensis, M. mytiloides and M. labiatus, and assumed only slight stratigraphic overlap between these species range zones. Subsequent high-resolution stratigraphic collection of the Early Turonian in Europe and America, however, revealed additional species and subspecies of Mytiloides, and rare Inoceramus (s.s.) in this interval, as well as more extensive stratigraphic overlap between some of the zonal species. Elder (1991) described M. hattini as a basal Turonian stratigraphic index; Kennedy et al. (1987) correctly pointed out that the type of Inoceramus opalensis Böse was distinct from Seitz's (1934) concept and was probably Middle to Late Turonian or even Coniacian in age, within the Mytiloides hercynicus and/or Inoceramus waltersdorfensis lineages. Subsequently, Early Turonian forms originally assigned to M. opalensis (Böse) by Seitz (1934) have been reassigned to M. kossmati Heinz (1933) by Walaszczyk (1992; confirmed in this workshop), who also synonymized M. goppelnensis Badillet and Sornay with M. kossmati. These observations, in turn, paved the way for simple assemblage zonation, and greater biostratigraphic refinement for the Early Turonian Mytiloides. The attempt of Kennedy and Cobban (1991) to simplify Early Turonian inoceramid zonation by establishing two zones (Mytiloides columbianus and M. mytiloides) for the section at Pueblo, Colorado, is herein rejected. Their species concepts are far broader than those utilized by specialists in the field (e.g., the Hamburg working group), and they have made serious taxonomic errors in the manner in which these names are utilized.
Whereas the Hamburg working group on Lower Turonian Inoceramidae discussed stratigraphic ranges for most latest Cenomanian and Lower to Middle Turonian species, no compilation of inoceramid biostratigraphy was attempted at the meeting, but it was noted that somewhat different zonal systems were utilized in Eurasia and North America, that some taxonomic changes were needed and species range zones modified, and that additions could be made to both systems. Subsequently, some of the workshop participants offered preliminary drafts of new or modified zonal schemes for the latest Cenomanian - Middle Turonian interval, and these are included herein as preliminary zonal hypotheses for future discussion and modification. Hilbrecht (November, 1993, personal communication) further suggested a simplified global zonal scheme for the Early and early Middle Turonian, utilizing inoceramids, of Mytiloides hattini (base), M. mytiloides, M. labiatus, and M. hercynicus.
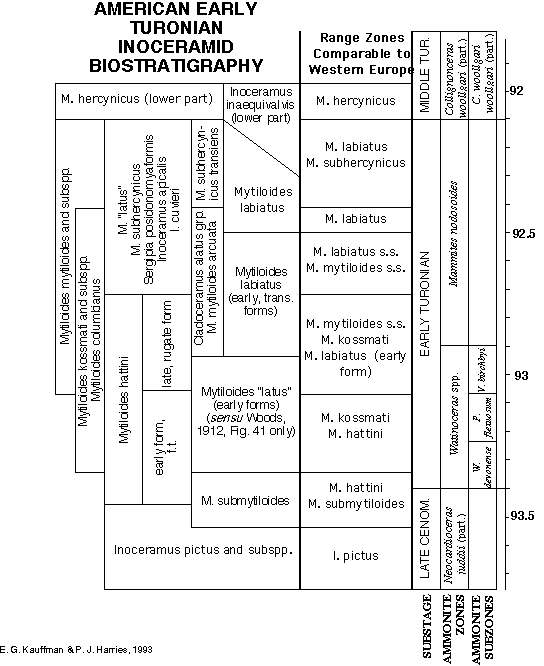
Figure 6 represents the most recent compilation of Lower Turonian inoceramid species and subspecies ranges for the North American Gulf Coast and Western Interior Cretaceous seaways, reflecting the composite opinions of the WIK Chronology working group of the Global Sedimentary Geology Program (GSGP: CRER). High-resolution stratigraphic data from North America has been primarily compiled by Kauffman (1975, 1977), Kauffman et al. (1976, 1993, in press), Elder (1987, 1989), and Harries (1990, 1993). These ranges have been established utilizing HIRES sampling techniques (Kauffman, 1988a; Kauffman et al., 1991), and involved continuous bulk sampling of 10-20 cm intervals across the Cenomanian-Turonian boundary at nearly 100 sections from Texas to southern Canada. Integration of range zone data was by linear and graphic correlation utilizing 112 event-chronostratigraphic marker horizons/beds (bentonites, Milankovitch climate cycle deposits, etc.). The distribution of composite inoceramid taxon range zones for North America is shown in Figure 6, in addition to the probable stratigraphic position of Brazilian Cladoceramus (= Rhyssomytiloides) from the Sergipe Basin. Note that a number of chronologically (evolutionarily) transitional forms between described species are recognized and plotted to give our colleagues a better view of the extent of each species plexus; some of these may receive new subspecies names during planned taxonomic revision. From this diagram, a series of assemblage biozones can be constructed, utilizing Inoceramidae, and tied to the standard ammonite zonation of Cobban (1985; 1993 in press) and Kennedy and Cobban (1991) for Euramerica. Figure 7 shows simple composite range zones (represented by the individual boxes) constructed from the stratigraphic ranges of the most abundant and widespread Lower Turonian inoceramids in America. These composite zones can be used for inter-regional correlation. Through the combination and overlap of these composite range zones, the Lower Turonian can be subdivided into eight different assemblage zones in this region.
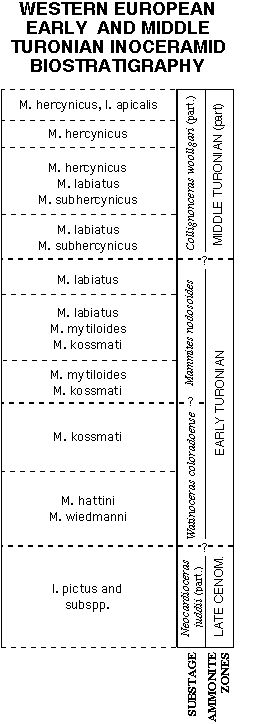
Figure 8 presents the latest inoceramid biozonation for the Western European Early to Middle Turonian by Walaszczyk (1992), as endorsed by Tröger (personal communication, Dec., 1993), and with the addition of Mytiloides hattini Elder and M. wiedmanni Lopez in the basalmost Turonian, as suggested by Hilbrecht (personal communication, November, 1993) and Lopez (personal communication, December, 1993), respectively. In his communication, Lopez further suggested inclusion of subspecies originally described under M. goppelnensis, and now valid subspecies of M. kossmati (see Walaszczyk, 1992, and previous discussion). But because the stratigraphic ranges of these subspecies are not yet well known, they have not been included in this preliminary European biozonation (Fig. 8).
At the broad scale of zonation, the North American and European biozones compare favorably among Inoceramidae. Both define basal Turonian zones characterized by typical Mytiloides hattini (regionally co-occurring with M. wiedmanni or M. submytiloides), a second biozone with M. kossmati (with or without M. hattini), a third concurrent range zone of M. kossmati with M. mytiloides, a fourth concurrent range zone of M. kossmati, M. mytiloides, and M. labiatus, a fifth biozone dominated by M. labiatus, and an late Early /early Middle Turonian biozone characterized by M. labiatus with M. subhercynicus, overlain by an M. hercynicus zone. Exact ammonite boundaries are more difficult to define in European successions due to the relative paucity of ammonites, and further refinement is required to pin-point the stage boundaries. Finer scale divisions of the North American sequence may reflect higher intensity of collecting, at closer stratigraphic levels, inherent in high-resolution stratigraphy, as well as the relatively less condensed nature of many North American Lower Turonian sequences.
CONCLUSIONS
The Lower Turonian Inoceramidae underwent a major evolutionary radiation following the Cenomanian-Turonian boundary mass extinction, primarily among members of the genus Mytiloides. Rapid evolutionary rates coupled with rapid intercontinental to cosmopolitan dispersal of many species present a perplexing evolutionary problem, but produces one of the most refined inter-regional biostratigraphic zonations of the Cretaceous. As with any major radiation, taxonomic problems abound, and phylogenetic relationships are sometimes difficult to define. Even among experienced workers in the field who attended the Hamburg workshop, no consensus could be reached on the phylogenetic relationships of Lower Turonian Inoceramidae. But the meeting did produce important advances in our understanding of this unique group of bivalves. Generic, and to a large degree, species concepts were stabilized, morphologic and morphometric parameters defined for future systematic work, the ecology and life habit of these Inoceramidae broadly discussed, and preliminary biostratigraphic revisions made for North America and Western Europe. Perhaps most valuable to all of us was the open forum for discussion and the identification of areas of controversy which will stimulate further work, and guide future workshops.
ACKNOWLEDGEMENTS: We would like to thank Prof. Christian Spaeth (Hamburg) for allowing us to include our workshop as part of the 4th International Cretaceous Symposium (Sept. 26-Oct.4, 1992), and we would also like to thank Heinz Hilbrecht (ETH, Switzerland) and Peter Harries (Tampa, FL, USA) for the exceptional job they did in organizing the workshop and making it a reality. The input of all of our colleagues at the workshop and the sharing of ideas aided greatly in this contribution.
REFERENCES CITEDAliev, M. M., Pavlova, M. M., Pergament, M. A., 1988, Standardization of terminology, ornament, and measurement of morphological elements of shells of Cretaceous inoceramids: In: Aliev, M. M. (ed.), Problems of Paleontology and Stratigraphy. Nauka, Moscow. [Posthumous collection of papers by M. M. Aliev, compiled by B. S. Sokolov, V. V. Menner, A.A. Ali-zade. In Russian], p. 17-45.
Badillet, G., and Sornay, J., 1980, Sur quelgues formes du grupe d'Inoceramus labiatus décrites par O. Seitz. Imposibilité d'utiliser ce grupe pour une datation stratigraphique du Turonien inférieur du Saumurois (France). Compte Rendu Academie Science Paris, Serie D, v.290, p. 323-325.
Bengtson, P., 1983, The Cenomanian - Coniacian of the Sergipe Basin, Brazil. Fossils and Strata, n. 12, p. 1-78.
Böhm, J., 1915, Vorlag von Inoceramen aus dem Subhercynen Emscher und Untersenon. Zeitschrift der deutschen geologischen Gesellschaft, v. 67, p. 181 - 183.
Brongniart, A., 1822, in Cuvier, G., Recherches sur les Ossemens Fossiles. Paris: G. Dufour et E. d'Ocagne; v. 2, pt. 2, p. 333-609.
Cobban, W. A. and Reeside, J. T., 1952, Correlation of the Cretaceous formations of the Western Interior of the United States. American Association of Petroleum Geologists Bulletin, v. 36, p. 1011-1044.
Cobban, W. A., 1985, Ammonite record from Bridge Creek Member of Greenhorn Limestone at Pueblo Reservoir State Recreation Area, Colorado: In L. M. Pratt, E. G. Kauffman and F. B. Zelt (eds.), Fine-grained Deposits and Biofacies of the Cretaceous Western Interior Seaway: Evidence of Cyclic Sedimentary Processes. Society of Economic Paleontologists and Mineralogists: 1985 Midyear Meeting, Golden, CO, Fieldtrip Guidebook 4, p. 135-138.
Cox, L. R., 1969, Family Inoceramidae Giebel, 1852: In Moore R. C. (ed.), Treatise on Invertebrate Paleontology, Part N, v. 1, Mollusca 6, Bivalvia. Lawrence, KS: University of Kansas Press and Geological Society of America, p. N314-N321.
Crampton, J. S., 1992, Morphometric description of cosmopolitan early Cretaceous inoceramid bivalves: In Lidgard, S. and Crane, P. R. (eds.), Fifth North American Paleontological Convention, Abstracts with Program. Paleontological Society Special Publication 6, p. 72 [Abstract].
Dhondt, A.V., and Dieni, I., 1990, Unusual inoceramid-spondylid association from the Cretaceous Scaglia Rossa of Passo del Brocon (Trento, N. Italy) and its palaeoecologoical significance. Memmorie degli Isituti di Geologia e Mineralogia dell'Università di Padova, v. 42, p. 155-187.
Dobrov, S. A., and Pavlova, M. M., 1959, Inoceramidae: In M. M. Moskvin (ed.), Atlas of the Upper Cretaceous fauna of northern Caucasus and Crimea. Trudy VNIIGAZ, p. 130-165 [In Russian].
Efremova, V. I., 1978, On methods and the unification of measurements of morphological elements of inoceram tests: In Pergament, M. A. (ed.), Jurassic and Cretaceous Inocerams and their Stratigraphic Importance. (Material of the III and IV All-Union Colloquium). Akademia Nauk SSSR, Geological Institute, Moscow, p. 99 - 104. [In Russian].
Einsele, G., Ricken, W., and Seilacher, A., 1991, Cycles and Events in Stratigraphy. Berlin: Springer-Verlag, 955 pp.
Elder, W. P., 1987, The Cenomanian-Turonian (Cretaceous) stage boundary extinctions in the Western Interior of the United States. Unpublished Ph. D. dissertation, Univ. of Colorado, 620 pp.
Elder, W. P., 1989, Molluscan extinction patterns across the Cenomanian-Turonian stage boundary in the Western Interior of the United States. Paleobiology, v. 15, p. 299-320.
Elder, W. P., 1991, Mytiloides hattini n. sp.: A guide fossil for the base of the Turonian in the Western Interior of North America. Journal of Paleontology, v. 65, p. 234-241.
Ferson, S., Rohlf, F. J., Koehn, R. K., 1985, Measuring shape variation of two-dimensional outlines. Systematic Zoology, v. 34, p. 59 - 68.
Foster, D. W. and Kaesler, R. L., 1988, Shape analysis; ideas from the Ostracoda: In McKinney, M. L. (ed.) Heterochrony in Evolution: A Multidisciplinary Approach. New York: Plenum Press, p. 53-69.
Goldfuss, A., 1836, Petrefacta Germaniae, 1833-1841; II. Teil. Düsseldorf: Arnz and Co., 312 pp.
Harries, P. J. and Kauffman, E. G., 1990, Patterns of survival and recovery following the Cenomanian-Turonian (Late Cretaceous) mass extinction in the Western Interior Basin, United States: In E. G. Kauffman and O. H. Walliser (eds.), Extinction Events in Earth History, Berlin, Springer-Verlag, Lecture Notes in Earth History 30, p. 277-298.
Harries, P. J., 1993, Pattern of repopulation following the Cenomanian-Turonian (Late Cretaceous) mass extinction. Unpublished Ph. D. dissertation, Univ. of Colorado, 356 pp.
Heinz, R., 1928, Das Inoceramen-Profil der Oberen Kreide Lüneburgs mit Anführung der neuen Formen und deren Kennzeichnung. Beiträge zur Kenntnis der oberkretazischen Inoceramen I. Jahresbericht des Niedersächsischen geologischen Vereins zu Hannover, v. 21, p. 63-81.
Heinz, R., 1932, Aus der neuen Systematik der Inoceramen (Inoceramen XIV). Mitteilungen aus dem mineralogisch-geologischen Staatsinstitut in Hamburg, v. 13, p. 1-26.
Heinz, R., 1933, Inoceramen von Madagaskar und ihre Bedeutung für die Kreide-Stratigraphie (Beiträge zur Kenntnis der Inoceramen XII). Zeitschrift der deutschen geologischen Gesellschaft, v. 85, p. 241-259.
Hessel, M. H. R., 1988, Lower Turonian inoceramids from Sergipe, Brazil: Systematics, stratigraphy and palaeoecology. Fossils and Strata, v. 22, p. 1-49.
Jablonski, D., 1986, Background and mass extinctions: The alternation of macroevolutionary regimes. Science, v. 231, p. 129-133.
Jones, B., 1988, Paleoscene 8. Biostatistics in paleontology. Geoscience Canada, v. 15, p. 3 - 22.
Kauffman, E. G., 1975, Dispersal and biostratigraphic potential of Cretaceous benthonic Bivalvia in the Western Interior: In Caldwell, W. G. E. (ed.), The Cretaceous System in the Western Interior of North America. Geological Association of Canada Special Paper 13, p. 163-194.
Kauffman, E. G., 1976a, British middle Cretaceous inoceramid biostratigraphy: In Thomel, G., and Reyment, R. (eds.), Evènements de la Partie Moyenne du Crétacé (Mid-Cretaceous Events): Uppsala-Nice Symposia, 1975-1976. Annales du Muséum d'Histoire Naturelle de Nice, v. 4, p.IV.1 - IV.12.
Kauffman, E. G., 1976b, An outline of middle Cretaceous marine history and inoceramid biostratigraphy in the Bohemian Basin, Czechoslovakia: In Thomel, G., and Reyment, R. (eds.), Evènements de la Partie Moyenne du Crétacé (Mid-Cretaceous Events): Uppsala-Nice Symposia, 1975-1976. Annales du Muséum d'Histoire Naturelle de Nice, v. 4, p. XIII.1 - XIII.12.
Kauffman, E. G., 1976c, South African Middle Cretaceous Inoceramidae: In Thomel, G. and Reyment, R. (eds.), Evènements de la Partie Moyenne du Crétacé (Mid-Cretaceous Events): Uppsala-Nice Symposia, 1975-1976. Annales du Muséum d'Histoire Naturelle de Nice, v. 4, p. XVII.1-XVII.6.
Kauffman, E. G., 1988a, Concepts and methods of high-resolution event stratigraphy. Annual Reviews of Earth and Planetary Sciences, p. 605-654.
Kauffman, E. G., 1988b, The case of the missing community: Low-oxygen adapted Paleozoic and Mesozoic bivalves ("flat-clams") and bacterial symbioses in typical Phanerozoic oceans. Geological Society of America, Abstracts with Programs, v. 20, p. A48.
Kauffman, E. G., 1991, Biostratigraphy: Inoceramids: In Larson, P. A., Morin, R. W., Kauffman, E. G., and Larson, A. (eds.), Sequence stratigraphy and cyclicity of lower Austin-upper Eagle Ford outcrops (Turonian-Coniacian), Dallas County, Texas. Dallas Geological Society, Field Trip #9 (Guidebook), p. 17-24.
Kauffman, E. G., Cobban, W. A., and Eicher, D. L., 1976, Albian through Lower Coniacian strata, biostratigraphy and principal events, Western Interior United States: In Thomel, G., and Reyment, R., (eds.), Evènements de la Partie Moyenne du Crétacé (Mid-Cretaceous Events): Uppsala-Nice Symposia, 1975-1976. Annales du Muséum d'Histoire Naturelle de Nice, v. 4, p. XXIII.1-XXIII.24.
Kauffman, E. G., Elder, W. P., and Sageman, B. B., 1991, High-resolution correlation: A new tool in chronostratigraphy: In Einsele, G., Ricken, W., and Seilacher, A. (eds.), Cycles and Events in Stratigraphy. Berlin: Springer-Verlag, p. 795-819.
Kauffman, E. G. and Harries, P. J., 1992, The ecology and life-habits of Cenomanian-Turonian Inoceramidae in North America. Workshop on Early Turonian Inoceramids (5-6 Oct, 1992, Hamburg) [Abstract].
Kauffman, E. G., and Powell, J. D., 1977, Part 2: Paleontology: In Kauffman, E. G., Hattin, D. E., and Powell, J. D. (eds.), Stratigraphic, paleontologic, and paleo-environmental analysis of the Upper Cretaceous rocks of Cimarron County, northwestern Oklahoma. Geological Society of America Memoir 149, p. 47-150.
Kauffman, E. G., and Runnegar, B., 1975, Atomodesma (Bivalvia) and Permian species from the United States. Journal of Paleontology, v. 49, n. 1, p. 23-41.
Kauffman, E. G. and Sageman, B. B., 1990, Biological sensing of benthic environments in dark shales and related oxygen-restricted facies: In R. N. Ginsberg and B. Beaudoin (eds.), Cretaceous Resources, Events and Rhythms: Background and Plans for Research, NATO ASI Series C: Mathematical and Physical Sciences, v. 304, p. 121-138.
Kauffman, E. G., Sageman, B. B., Kirkland, J. I., Harries, P. J., and Villamil, T., 1993 in press, Molluscan biostratigraphy of the Western Interior Cretaceous Basin, North America: In Caldwell, W. G. E., and Kauffman, E. G. (eds.), Mesozoic Evolution of the Western Interior Basin of North America: Geological Association of Canada, Special Paper.
Keller, S., 1982, Die Oberkreide der Sack-Mulde bei Alfeld (Cenoman-Unter Coniac): Lithologie, Biostratigraphie und Inoceramen. Geologisches Jahrbuch, v. A96, 185 pp.
Kennedy, W. J., Wright, C. W., and Hancock, J. M. 1987, Basal Turonian ammonites from West Texas. Palaeontology, v. 30, p. 27-74.
Kennedy, W. J. and Cobban, W. A. 1991, Stratigraphy and interregional correlation of the Cenomanian-Turonian transition in the Western Interior of the United States near Pueblo, Colorado, a potential boundary stratotype for the base of the Turonian stage. Newsletters on Stratigraphy, v. 24, 1-33.
Lopez, G., 1986, Inocerámidos del Cretácio superior de los alrededores de St. Corneli (Prov. Lleida). Publicaciones de Geologia Universitat Autónoma de Barcelona, v. 22, p. 1-123.
Lopez G., 1992a, Paleontología y Bioestratigrafía de los inocerámidos Bivalvia del Cretácio superior de la Cuenca Navarro-Cántabra y de la Platafroma Norcastellana. Parte II: Estudio sistemático de los subgéneros Mytiloides Brongiart y Magadiceramus Seitz. Boletín Geológico y Minero, v. 103 (3), p. 70-135.
Lopez G., 1992b, Paleontología y Bioestratigrafía de los inocerámidos Bivalvia del Cretácio superior de la Cuenca Navarro-Cántabra y de la Platafroma Norcastellana. Parte III: Estudio sistemático de los subgénero Platyceramus Seitz. Boletín Geológico y Minero, v. 103 (4), p. 23-81.
MacLeod, K. G. and Hoppe, K. A., 1992, Evidence that inoceramid bivalves were benthic and harbored chemosynthetic symbionts. Geology, v. 20, p. 117-120.
Matsumoto, T., Noda, M., and Kozai, T., 1982, Upper Cretaceous inoceramids from the Monobe area, Shikoku. In T. Matsumoto and M. Tashiro (eds.), Multidisciplinary Research in the Upper Cretaceous of the Monobe Area, Shikoku, Palaeontological Society of Japan, Special Papers, v. 25, p. 53-68.
Maury, C. J., 1925, Fosseis terciarios do Brasil, com des crião de novas formas cretaceas. Servico Geologico e Mineralogico do Brasil, Monographia 4, p. 1-705.
Noda, M., 1975, Succession of Inoceramus in the Upper Cretaceous of Southwest Japan. Memoirs of the Faculty of Science, Kyushu University, series D, geology, v. 23, p. 211-261.
Noda, M., 1983, Notes on the so-called Inoceramus japonicus (Bivalvia) from the Upper Cretaceous of Japan. Transaction and Proceedings of the Palaeontological Society of Japan, N. S., v. 158, p. 191-219.
Noda, M., 1988, A note on Inoceramus tenuistriatus Nagao et. Matsumoto (Bivalvia) from the Upper Turonian (Cretaceous) of Japan. Transactions and Proceedings of the Palaeontological Society of Japan, New Series, v. 151, p. 582 - 600.
Rasemann, W., 1986, Eine Klassifikation der Merkmale der Inoceramenschale. Freiberger Forschungshefte, Geowissenschaften - Palaeontologie, v. C 410, p. 45 - 58.
Rohlf, F. J. and Archie, J. W., 1984, A comparison of Fourier methods for the description of wing shape in mosquitoes (Diptera: Culicidae). Systematic Zoology, v. 3, p. 302 - 317.
Rohlf, F.J. and Bookstein, F. L. (eds), 1990, Proceedings of the Michigan Morphometrics Workshop. Ann Arbor, Michigan, University of Michigan , Museum of Zoology, Special Publication 2. Viii+380 pp.
Sageman, B. B., 1989, The benthic boundary biofacies model: Hartland Shale Member, Greenhorn Formation (Cenomanian), Western Interior, North America. Palaeogeograhy, Palaeoclimatology, Palaeoecology, v. 74, p. 87-110.
Schlüter, C., 1877, Kreide-Bivalven; Zur Gattung Inoceramus. Palaeontographica, v. 24, N.F. 4 (6), p. 250-289.
Scott, G. H., 1980, The value of outline processing in the biometry and systematics of fossils. Palaeontology, v. 23, p. 757 - 768.
Seitz, O., 1934, Die Variabilität des Inoceramus labiatus v. Schlotheim. Jahrbuch der Königlichen Preussischen Geologischen Landesanstalt, v. 55 (1), p. 426-274.
Seitz, O., 1961, Die Inoceramen des Santon von Nordwestdeutschland: I. Teil (Die Untergattungen Platyceramus, Cladoceramus, und Cordiceramus). Beihefte zum Geologischen Jahrbuch, v. 46, 186 pp.
Seitz, O., 1965, Die Inoceramen des Santon und Unter-Campan von Nordwestdeutschland: II. Teil (Biometrie, Dimorphismus und Stratigraphie der Untergattung Sphenoceramus J. Bohm). Beihefte zum Geologischen Jahrbuch, v. 69, 194 pp.
Seitz, O. 1967, Die Inoceramen des Santon und Unter-Campan von Nordwestdeutschland: III. Teil (Taxonomie und Stratigraphie der Untergattungen Endocostea, Haenleinea, Platyceramus, Cladoceramus, Selenoceramus und Cordiceramus, mit besonderer Berücksichtigung des Parasitismus bei diesen Untergattungen). Beihefte zum Geologischen Jahrbuch, v. 75, 171 pp.
Sornay, J., 1966, Idées actuelles sur les inocérames d'apres divers travaux recents. Annales de Paléontologie (Invertébrés), v. 52, p. 57 - 92.
Stanley, S. M., 1979, Macroevolution: Patterns and Process. San Francisco, W. H. Freeman, 332 pp.
Tanabe, K., 1973, Evolution and mode of life of Inoceramus (Sphenoceramus) naumanni Yokoyama emend, an Upper Cretaceous bivalve. Transactions and Proceedings of the Paleontological Society of Japan, New Series, v. 92, p. 163 - 184.
Temple, J. T., 1992, The progress of quantitative methods in paleontology. Palaeontology, v. 35, p. 475 - 484.
Tröger, K.-A., 1967, Zur Paläontologie, Biostratigraphie und faziellen Ausbildung der unteren Oberkreide (Cenoman bis Turon). Teil I. Paläontologie und Biostratigraphie der Inoceramen des Cenomans bis Turons Mitteleuropas. Abhandlungen des Staatlichen Museums für Mineralogie und Geologie zu Dresden, v. 12, p. 13-207.
Tröger, K.-A., 1986, Inoceramus (Mytiloides) incertus prescheri n. ssp., eine neue Unterart der Inoceramus-incertus-Gruppe aus dem Oberturon von Dresden-Strehlen. Freiberger Forschungshefte, Geowissenschaften - Palaeontologie, v. C410, p. 41 - 43.
Walaszczyk, I., 1992, Turonian through Santonian deposits of the Central Polish Uplands; their facies development, inoceramid paleontology and stratigraphy. Acta Geologica Polonica, v. 42, p. 1-122.
White, R. J., Prentice, H. C., and Verwijst, J., 1988, Automated image acquisition and morphometric description. Canadian Journal of Botany, v. 66, p. 450 - 459.
Woods, H., 1912, A monograph of the Cretaceous Lamellibranchia of England: Volume 2, Part 8, Inoceramus. Monograph of the Palaeontolographical Society (for 1911), p. 285-340.
Vialov, O. S., 1960, Family Inoceramidae Heinz, 1932: In Eberzin, A. G. (ed.), Paleontologsy Osnovy; Spravochnik dlya Paleontologov i Geologov SSSR: Mollyuski. Izdatel'stvo. Akad. Nauk SSSR, Moscow. 303 pp.